 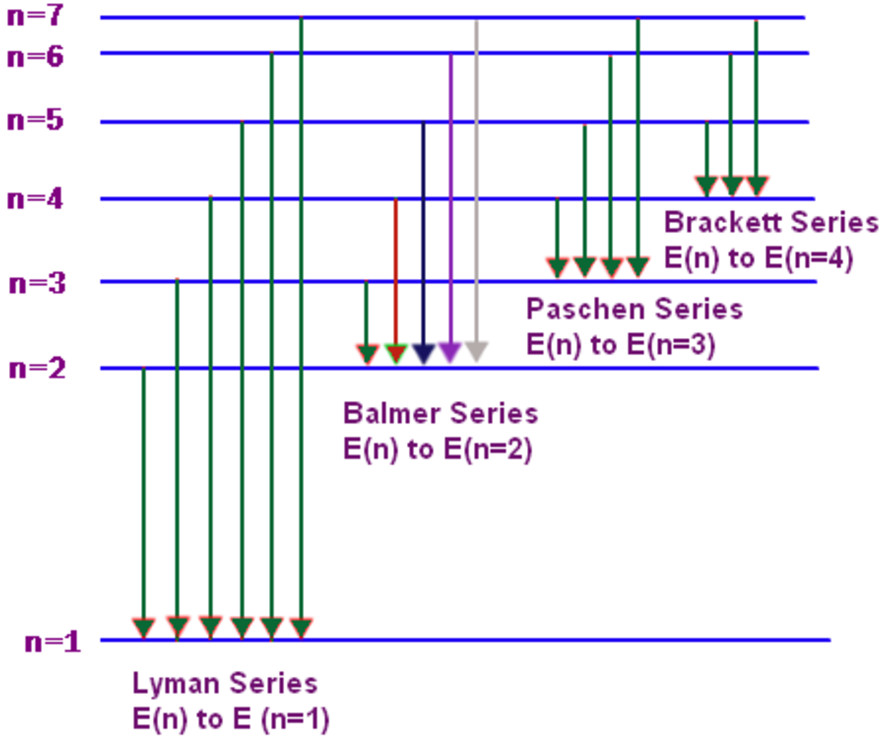
Those two energy levels are that difference in energy is equal to the energy of the photon. So an electron is falling from n is equal to three energy levelĭown to n is equal to two, and the difference in To n is equal to two, I'm gonna go ahead andĭraw an electron here. All right, so if an electron is falling from n is equal to three Now let's see if we can calculate the wavelength of light that's emitted. All right, so it's going to emit light when it undergoes that transition. So we have an electron that's falling from n is equal to three down to a lower energy level, n is equal to two. For example, let's say we were considering an excited electron that's falling from a higher energy Where I is talking about the lower energy level, minus one over J squared, where J is referring to the higher energy level. The Rydberg constant, times one over I squared, Of light that's emitted, is equal to R, which is And you can see that one over lamda, lamda is the wavelength So I call this equation theīalmer Rydberg equation. And we can do that by using the equation we derived in the previous video. And since line spectrum are unique, this is pretty important to explain where those wavelengths come from. Is unique to hydrogen and so this is one way A blue line, 434 nanometers, and a violet line at 410 nanometers. You'll also see a blue green line and so this has a wave 
So you see one red lineĪnd it turns out that that red line has a wave length. So this is the line spectrum for hydrogen. So, since you see lines, weĬall this a line spectrum. If you use something likeĪ prism or diffraction grating to separate out the light, for hydrogen, you don't When those electrons fallĭown to a lower energy level they emit light and so we talked about this in the last video. So, if you passed a current through a tube containing hydrogen gas, the electrons in the hydrogen atoms are going to absorb energy and jump up to a higher energy level. Thing with hydrogen, you don't see a continuous spectrum. So that's a continuous spectrum If you did this similar It's continuous because you see all these colors right next to each other. Like this rectangle up here so all of these differentĬolors of the rainbow and I'm gonna call thisĪ continuous spectrum. And so if you did this experiment, you might see something Of light through a prism and the prism separated the white light into all the differentĬolors of the rainbow. I'm sure that most of you know the famous story of Isaac Newton where he took a narrow beam of light and he put that narrow beam 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
